On today’s stock market, Eli Lilly stock surged 6.7% to close at 431.19. Eli Lilly’s market cap closed above $400 billion for the first time ever, hitting roughly $409 billion.

Eli Lilly (LLY) stock blazed higher Wednesday after the company said its experimental Alzheimer’s treatment significantly slowed cognitive decline in people with an early stage of the disease.
On a scale that measures cognition, patients who received Lilly’s donanemab had a 35% slower decline vs. the placebo recipients. On a measurement of activities of daily living, donanemab patients showed a 40% slower decline compared with the placebo group.
But there are caveats in the data. Those numbers pertain only to patients with intermediate levels of a protein called tau in the brain. Tau is associated with Alzheimer’s disease. Across all patients, including those with high levels of tau, donanemab led to a 22% slower decline in cognition on the same scale.
“Overall, we’d characterize the results as ‘overhang removed,’ a ‘best-case scenario,’ especially as Lilly plans to file donanemab for regulatory approval this quarter,” Bank of America analyst Geoff Meacham said in a note to clients.
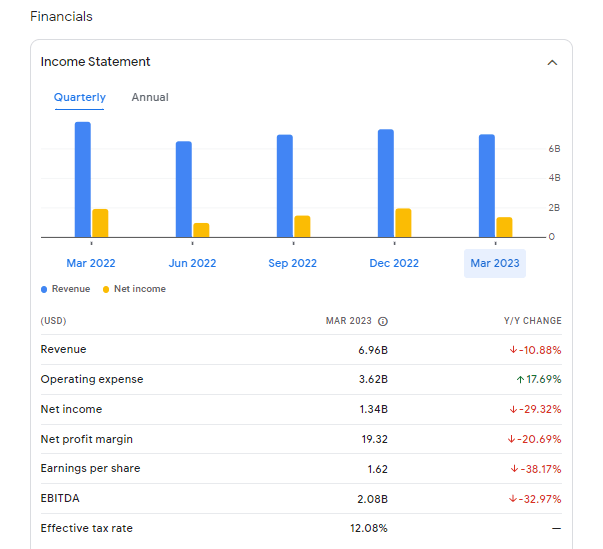
Eli Lilly and Company is an American pharmaceutical company headquartered in Indianapolis, Indiana, with offices in 18 countries. Its products are sold in approximately 125 countries. The company was founded in 1876 by, and named after, Colonel Eli Lilly, a pharmaceutical chemist and veteran of the American Civil War. As of 2022, Lilly is known for its clinical depression drugs Prozac, Cymbalta, and its antipsychotic medication Zyprexa, although its primary revenue drivers are the diabetes drugs Humalog and Trulicity. Lilly’s achievements include being the first company to mass-produce the polio vaccine developed by Jonas Salk, and insulin. It was one of the first pharmaceutical companies to produce human insulin using recombinant DNA including Humulin, Humalog, and the first approved biosimilar insulin product in the US, Basaglar. In 2009 Lilly pleaded guilty for illegally marketing Zyprexa and agreed to pay a $1.415 billion penalty that included criminal fine of $515 million, the largest ever in a healthcare case and the largest criminal fine for an individual corporation ever imposed in a US criminal prosecution of any kind at the time.
Eli Lilly Stock: Rivaling Biogen
Analysts were quick to compare donanemab to Biogen‘s (BIIB) approved Alzheimer’s treatment, Leqembi. Biogen used a different scale, called Clinical Dementia Rating-Sum of Boxes, or CDR-SB. Leqembi led to a 27% slower decline in cognition across all patients over 18 months.
In Lilly’s study, donanemab recipients with intermediate levels of tau had a 36% slower cognitive decline as measured by CDR-SB. But when accounting for all patients, including those with high tau levels, the experimental Alzheimer’s treatment led to a 29% slower decline in cognition vs. the placebo. Analysts noted that’s more in line with Biogen’s results.
Across all measurements, donanemab appeared to narrowly outperform Leqembi, RBC Capital Markets analyst Brian Abrahams said in a report. However, it’s important to note Biogen didn’t separate patients based on levels of tau in the brain.
Safety In Alzheimer’s Treatment
Further, there appear to be safety trade-offs with donanemab. Both donanemab and Leqembi work by removing a protein in the brain called amyloid. This can lead to brain swelling known as amyloid-related imaging abnormalities, or ARIA.
In Lilly’s study, temporary swelling occurred in 24% of donanemab recipients, and 6.1% of those reported symptoms. Brain bleeding, another form of ARIA, occurred in 31.4% of donanemab patients. In Biogen’s study, 12.6% of patients had brain swelling, including 2.8% with symptoms. Another 17.3% of patients experienced brain bleeding, William Blair analyst Myles Minter said in a note.
Notably, three patients died due to or after experiencing serious ARIA in Lilly’s study. In Biogen’s Phase 2 study, none of the patients died due to ARIA. In a final-phase study, two Leqembi recipients died, but the deaths weren’t deemed to be ARIA-related, Evercore ISI analyst Umer Raffat said in a note.
He has an in-line rating on Eli Lilly stock.
“In our view, this important safety differential is enough to distinguish these two drugs with a clear Leqembi advantage,” Piper Sandler analyst Christopher Raymond said in a report.
Raymond has an overweight rating on Biogen stock.
Biogen Stock Claws Back
Biogen stock initially toppled in premarket trading on the news, but clawed back to gain 0.6%, closing at 311.
Leqembi has accelerated approval, but Biogen is seeking a traditional approval based on a confirmatory study. This could allow the Centers for Medicare and Medicaid Services to reimburse beneficiaries for the Alzheimer’s treatment.
William Blair’s Minter says Lilly’s data could help in that pursuit.
“We continue to believe the positive (Lilly) data alongside the (Biogen) trial bodes well to support broader CMS reimbursement for the anti-amyloid class,” he said. He noted the FDA is due to make a decision on the full approval of Leqembi on July 6. CMS could make its reimbursement call around that time.
Minter has an outperform rating on Biogen stock.
Biogen shares popped out of a buy zone on Wednesday’s release. The biotech stock broke out of a double-bottom base with a buy point at 296 last month, according to MarketSmith.com. Eli Lilly stock is trading at fresh highs after overtaking a cup base entry at 375.35 in mid-April.

